Short version:
Inorganic fertilisers have changed the world but maybe now we need to consider the impact of their use and production. Organic fertilisers may be slower to work but they don’t consume as much energy in their production and can be less harmful. Consider composting and practises that help with soil health.
Why are fertilisers required?
Plants need 13 minerals in order to thrive. Of these nitrogen, phosphorus and potassium are needed in large quantities and sulphur, magnesium and calcium are need in lesser quantities. The other 7 are needed in very small amounts.
With intensive farming methods that include single crop fields, growing multiple crops per year and mechanised agriculture there is a very large output of cheap food produced per acre of land. These techniques strip the soil of nutrients. Once a plant has taken up nutrients out of the soil and then that plant is removed from the area during harvesting, those nutrients need to be replaced for the next crops to be grown. This means that there is a heavy reliance on fertilisers.
It was in the early twentieth century that the Haber process was developed. This is an industrial process that converts nitrogen from the air into ammonia (NH3), which can then be converted into a form that can be utilised by plants (nitrates NO3–). The nitrogen cycle can explain how plants get their nitrogen in nature.
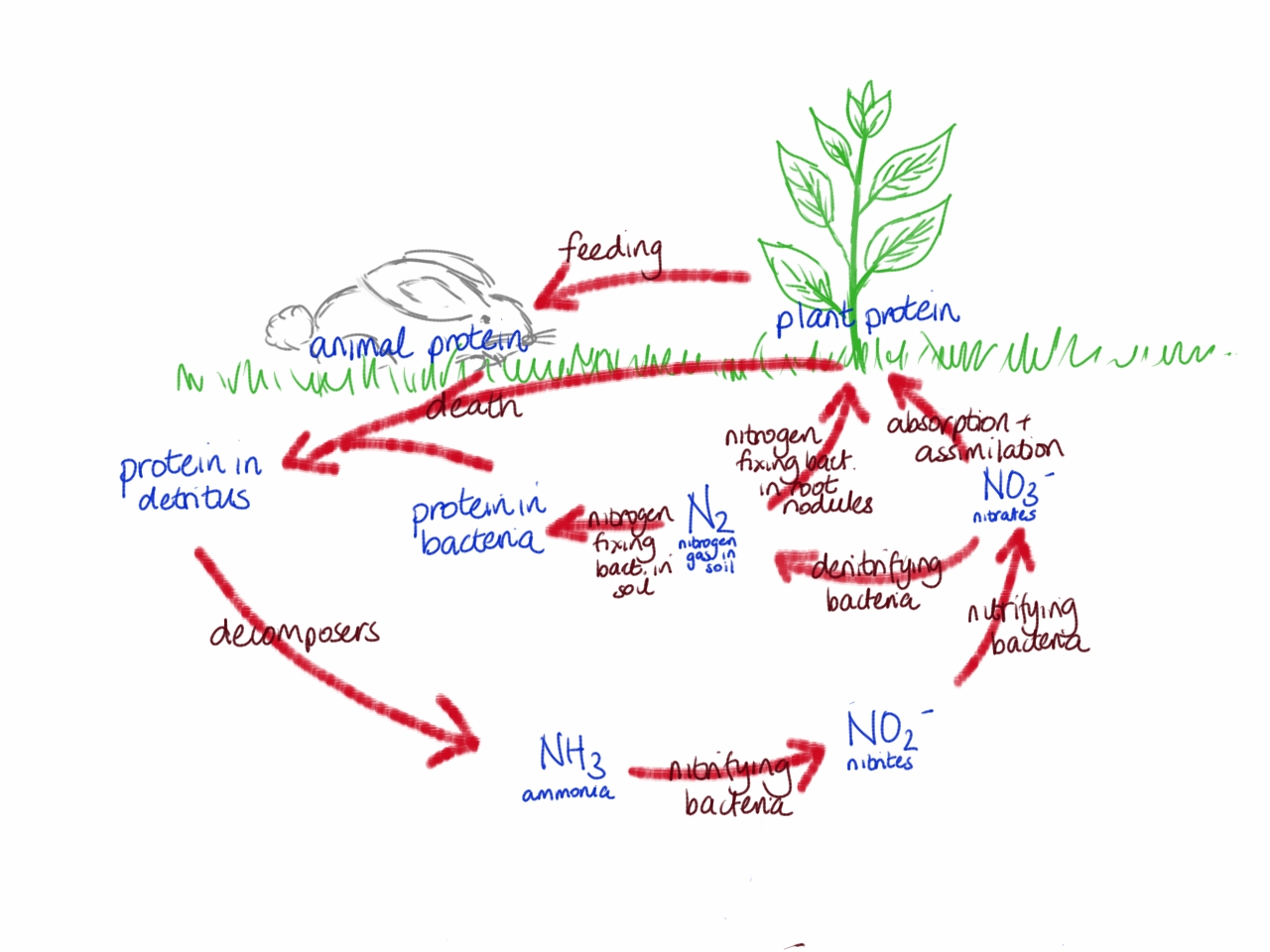
The Haber process allowed production of cheap inorganic fertilisers, heavy in nitrogen, which is the most required nutrient. This meant that it was now possible for food production to keep up with, and support rapidly increasing populations.
Basically, we’re now kinda scuppered! In order to keep producing enough food at prices that people can afford, and allow farms to continue to run, farmers have no choice but to keep using inorganic fertilisers and to keep using methods that aren’t particularly good for the soil.
However, in a domestic setting we have choices. I hope that you choose to limit your use of inorganic fertilisers. Why? Let’s start with how they’re made.
What is the Haber process?
At around 450oC and 200atm (high pressure), with an iron catalyst (which helps to speed up the reaction), hydrogen and nitrogen are combined to form ammonia.
Now, there a couple of things to consider. The hydrogen comes from reacting methane with steam at around 1000oC. The methane will primarily come from natural gas, so we have the same environmental issues that we have from the extraction of fossil fuels. To get nitrogen, air (which is made up of approx. 78% nitrogen) is cooled to around -200oC to condense all the different gases in the air and then allowed to warm to the boiling point of nitrogen. The nitrogen gas is then collected and recondensed for storage and transport.
I write around 450oC and 1000oC because it can differ depending on the manufacturer. These high pressures and temperatures, as well as -200oC require a large amount of energy to achieve. If any of the energy is derived from fossil fuels, then that energy requirement has led to the release of pollutants and greenhouse gases. Hydrogen can be made from the electrolysis of water (splitting water using electricity). This also requires a lot of energy. As we move towards using a higher percentage of renewable energy sources these issues become less. The problem is that hydrogen still and will (until technology moves on significantly) mostly come from methane.
Ammonia can be oxidised to nitric acid, which can then be converted to nitrates. Ammonia can also be reacted with acids to give ammonium salts. Both nitrates and ammonium salts are used as fertilisers. If ammonia and nitric acid are reacted, you get ammonium nitrate, a popular fertiliser. Other widely used fertilisers are potassium nitrate or ammonium phosphate, both of which require ammonia.
Another issue is that the uptake of inorganic fertilisers by plants is too slow to avoid fertilisers being washed away into bodies of water. Here is the info on why that is a problem in ‘Eutrophication.’
The one undeniable bonus of inorganic fertilisers is that they do allow for quick treatment of any nutrient deficiencies.
Organic fertilisers
There are fertilisers made using by products like blood and bone meal from slaughterhouses, or poultry manure or fish bone and blood. This might be an issue if you’re vegan so maybe seaweed, comfrey or nettle feeds might be more up your street. Organic fertilisers tend to be more expensive, but they take some breaking down and so are less soluble and don’t get washed away as much. With organic fertilisers you should check the labels for sterilisation but reputable providers should be fine.
An organic fertiliser that is high in nitrogen and free is urine. My granny used to use it quite a bit back in the day to grow fantastic greens. You do need to dilute it, something like 9 parts water to 1 part urine, or it will cause fertiliser burn (taking a tinkle straight into your garden is going to cause more harm than good). You might also want to water the roots and avoid getting it on the leaves of plants that you eat, especially salads. You might also want to wash your vegetables thoroughly when you pick. You also should probably avoid using it when you’re ill or taking medication. You may also just want to avoid it all together if you just think it’s too icky. I’m a little OCD so I find it hard to put urine into my watering can to dilute it. What I did do instead was wash the potty out into the garden during potty training. Post wee, I would fill the potty to the top with water and pour it into the garden. I’d then fill it again to rinse and pour that out into the garden too. Sometimes maybe I’d give a second rinse too, just because I have issues.
Composting and practices for better soil health
I still try and limit my use of fertilisers at all. Mostly because of the above reasons and partly because they cost money and need to be stored. Fertilisers can also cause burn if overapplied or applied to wet leaves and I’m not particularly good at following exact directions.
Instead I like to compost all my vegetable kitchen scraps from both garden grown and shop bought fruit and vegetables. Things like banana skins are high in potassium and it is said that a lot of the nutrients in fruit and veg is in the peel. So if you don’t like eating the peels of things (here we don’t tend to eat potato, carrot, avocado, banana, pomegranate skins to name but a few) you can use this to replace the nutrients that are coming out of the garden. Compost also helps to improve the soil and help its structure, especially as my soil is heavy clay. Compost is good for soil health and feeds the microorganisms and worms in the soil. It also helps with both water retention and drainage. It’s also free and is good for the environment as it means less waste going into landfill or needs to be collected and transported for processing.
Other things that make a difference is to grow lots of perennials. Because it is not the whole plant that is being removed yearly, there is less being taken out of the soil. With the decomposition of dead roots and leaves, nutrients are returned to the soil. With perennials you don’t dig into the soil so much so the little ecosystems around the plant get to thrive. The roots will also help against soil erosion.
If anyone has any other ideas that can help fertilise the garden in a cheap, eco-friendly way, your comments would be appreciated.
5 thoughts on “Fertilisers”